IRE Ablation Device
Project Overview
Evaluation of numerous electrode configurations for performing IRE Ablation Procedure. Electrode spacing and sizing tested using Comsol cell death modeling to optimize design. Preliminary modeling of catheter based design performed.
Project Objective
Develop an optimal electrode configuration for performing IRE Ablation along a 2 cm section of vessel wall. The design must fit within a 3 French catheter.
Specifications
- Evenly ablate 2 cm section of cell wall
- Target cells have 3 - 7mm diameters
- Wall thickness is 25% of cell diameter
- Vessel wall conductivity 0.271 S/m
- Muscle Conductivity 0.202 S/m
- Blood conductivity 0.7 S/m
Technical Process
1. Establish Cell Death Model
Created a FEA model within COMSOL that evaluated cell death based on input current, voltage, and electron configuration. Model established using biological conductivities sourced from existing research.
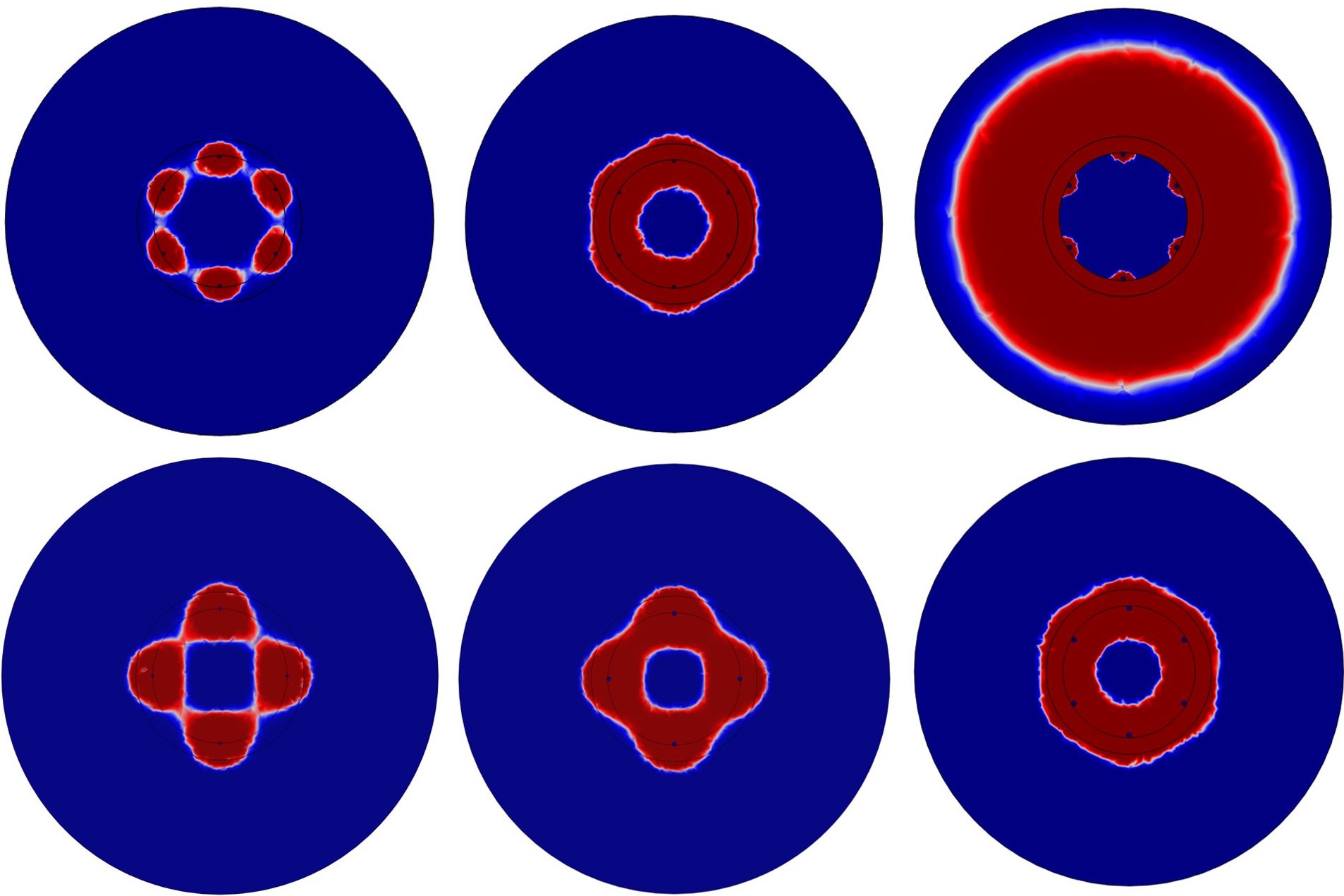
2. Evaluate Monopolar vs Bipolar Configuration
Utilizing custom cell death model, tested monopolar and bipolar electrode configuration for 60 and 80 pulses. Established that bipolar configuration resulted in more uniform ablation.
Monopolar - 60 Pulses 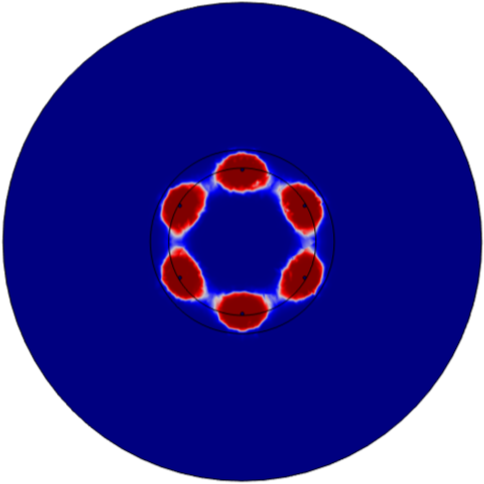
Bipolar - 60 Pulses 
Monopolar - 80 Pulses 
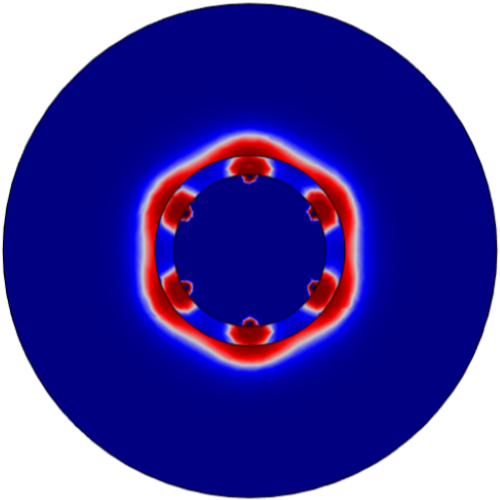
Bipolar - 80 Pulses 3. Evaluate Number of Electrodes
Compared 4, 6, and 8 electrode bipolar configurations with optimized electron diameters. Discovered 6 electrodes was optimal for ablating the entire vessel wall without additional cell death.

4 Electrodes 
6 Electrodes 
8 Electrodes 4. Optimize Number of Pulses
Tuned number of pulses for 6 electrode bipolar configuration at 3, 5, and 7mm vessel diameters.

3mm Vessel Diameter 
5mm Vessel Diameter 
7mm Vessel Diameter 5. Create Preliminary Catheter Design
Designed and modeled catheter to house electrodes and expand them to different vessel diameters. Electrodes intended to rest in buckled state and expand outwards as cover is retracted.

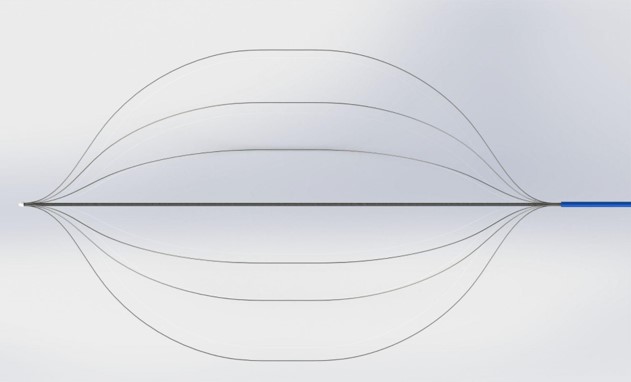

6. Conduct Competitor Research
Abbott Xience Stent technology investigated and regulatory pathway taken explored to lay framework for developing a marketable product.
Outcome and Impact
- Designed catheter based IRE ablation device optimized for achieving best results for target blood vessels.
- Gained valuable experience in using COMSOL software.
- Developed understanding of regulatory pathway required to take FDA Class 3 medical device to consumer market.