Catheter Hub w/ Hemostatic Lock

Project Overview
Design and prototyping of catheter hub that enabled the injection of contrast while immobilizing guidewire with hemostatic lock with adherence to FDA guidelines. Physical prototype manufactured and tested.
Project Objective
Design a prototype of a catheter hub that enabled the injection of contrast while immobilizing the guide wire in a hemostatic lock in accordance with ISO 594 and ISO/ASTM 52911-1 while adhering to the design process specified as per FDA 21.CFR 820.30.
Specifications
- Maintain Design History File throughout design and development process.
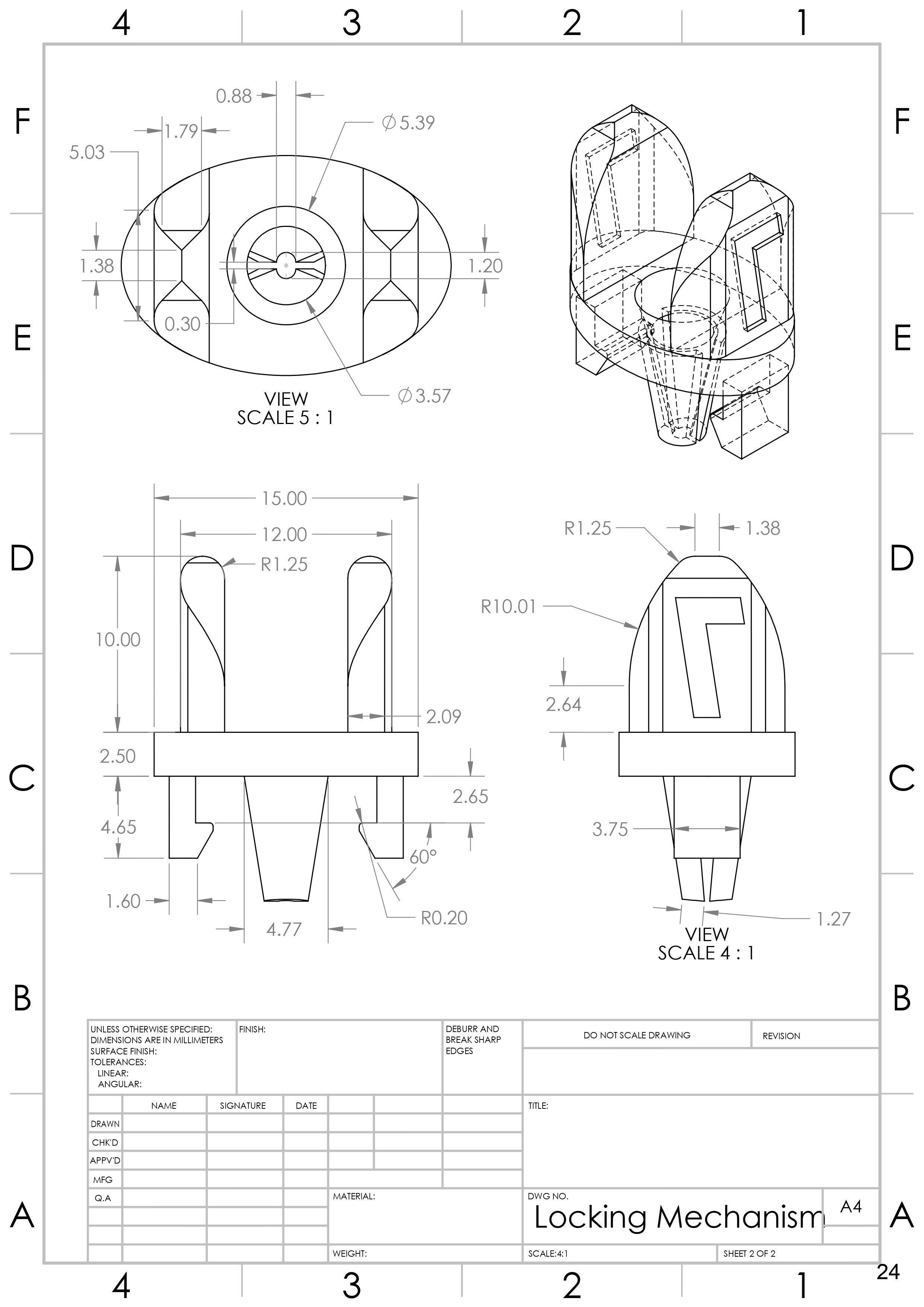
- Hemostatic lock with ability to immobilize guide wire.
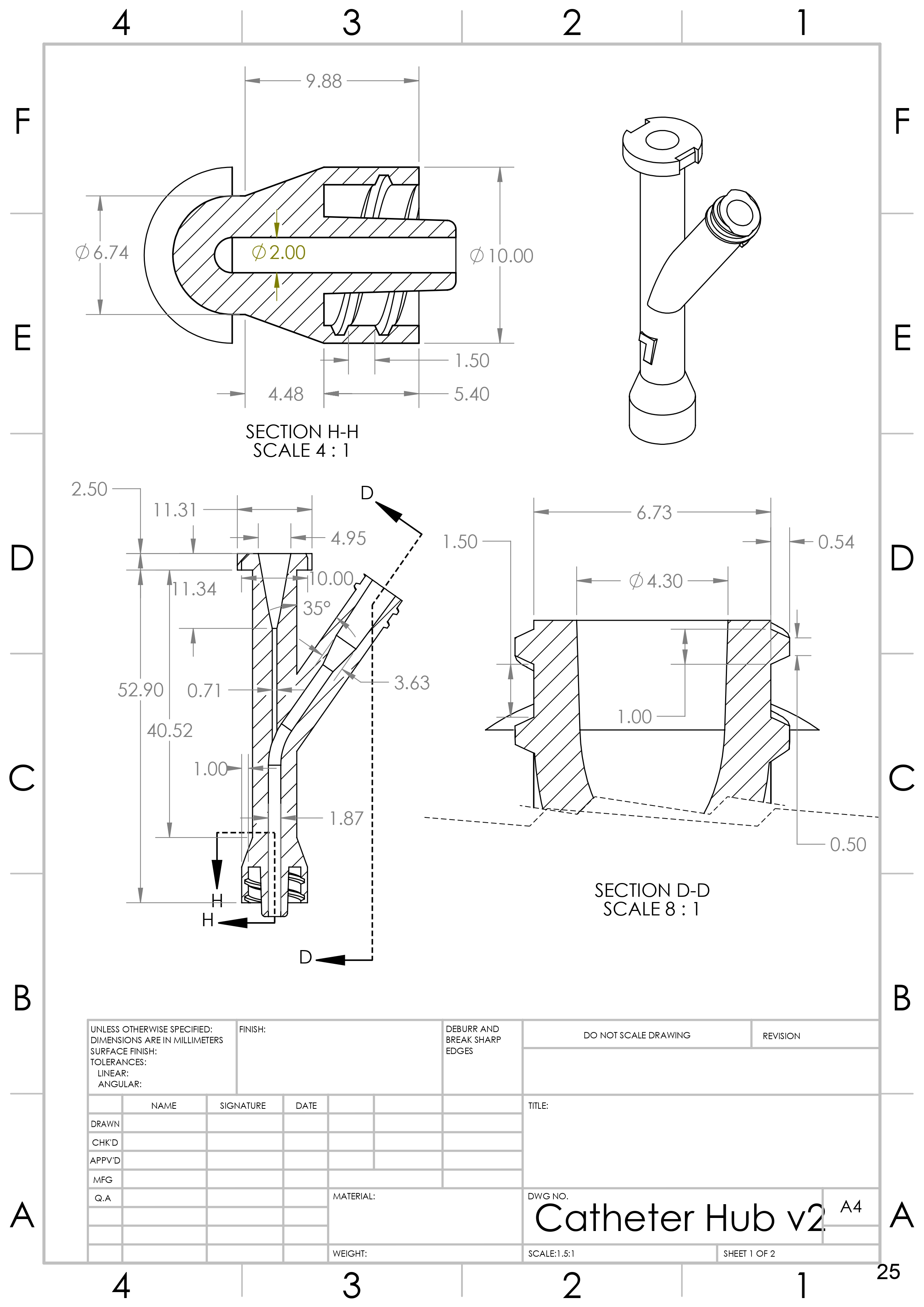
- ISO 594 luer lock that permits contrast injection.
- Design for fabrication with SLA 3D Printing.
Technical Process
1. Generate Design Requirements Matrix
Identified and categorized customer requirements and market specifications. Attributed rationale, performance expectation, source and test method to each specification.
2. Design Hemostatic Catheter Hub
Generated concepts for hemostatic locking catheter hub. Created 3D Model of selected catheter hub design with ISO 594 compatible male luer lock in SolidWorks.
3. SLA 3D Print and Post Process
3D printed catheter out of resin. Performed prototype post processing and recorded procedure.
4. Detail Testing Procedure and Perform Testing
Defined high pressure liquid leakage testing procedure based on 594-1 and a custom low pressure liquid leakage procedure. Conducted testing in accordance with written procedures and recorded outcomes.
5. Analyze Failure Modes/Cause of Failure
Analyzed failure modes of prototype and speculated on reasons design failed to achieve results called for in requirements matrix.
Outcome and Impact
- Gained experience in design practices called for in FDA 21.CFR 820.30 including maintaining Design History Files
- Learned to test in accordance with ISO 594-1
